New Submissions Are Not Currently Being Accepted
Samples sent after April 10, 2024 will be put on hold.
Viewing desktop version: Switch to Mobile
Purple Tablet (UPJOHN 90)
Sold as: MDMA / Ecstasy
ID: 18881
| ID: | 18881 |
|---|---|
| Name: | Purple Tablet |
| Other Names: | UPJOHN 90 |
| HPLC: |
|
| Test Date: | Jan 06, 2024 |
|---|---|
| Pub. Date: | Jan 23, 2024 |
| Src Location: | Zurich, Switzerland |
| Submitter Loc: | Zurich, -- Switzerland |
| Color: | Purple |
| Size: | 132.2 mg, 9.20 mm x 5.60 mm x 3.6 mm |
| Data Source: | SaferParty.ch |
| Tested by: | DIZ |
| Lab's ID: | SP:202401-31 |
Sold as: MDMA / Ecstasy
Expected to be: Not Specified

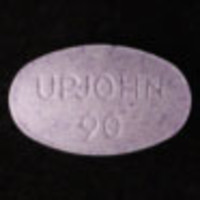
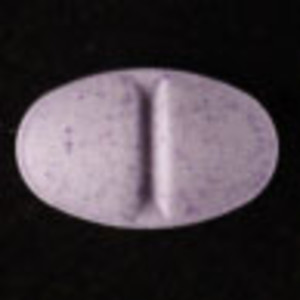

Analyzed by SaferParty (Switzerland) in January 2024.